Peptides Guide
Peptides play significant role in cell signaling and function and can be used as an important tool for research and therapeutic treatments. Here we discuss both the structure and activities of peptides as well as strategies for their synthesis.
What are peptides?
Both peptides and proteins fall under a category called polypeptide chains. These chains contain two or more amino acids (forming amino acid polymers) that are coupled by a peptide bond. The bond is a special linkage between the nitrogen atom of one amino acid and the carboxyl carbon atom of another.
Peptides differ from proteins by amount of amino acid residues the molecule contains. Polymer molecules with ten or fewer amino acid residues are called oligopeptides. Peptides often contain up to fifty amino acid residues, protein are molecules with more than fifty amino acid residues.
Peptides are functioning in human body on many ways, such as regulating metabolism (insulin) and mediating pain signals (dynorphin). They also play a role in endocrine signaling and can act as a growth factor. Other organisms have produced peptides as a means for defense, such as fungal production of cyclosporin A used clinically as an immunosuppressant, and cone snail secretion of Ziconotide which is used to treat a pain.
Why are they important?
Modern medicinal and biochemical research is unthinkable without peptides application because of their selectivity, specificity and potency interaction with the target proteins. Peptides' large size and surface area allow for more specific docking to the target molecules. Researchers' interest in developing peptide ligands and probes for studying target receptors' structures and functions has increased dramatically lately.
More recently, peptides have been considered as the desirable candidates for therapeutics. Not only can peptides be made very selelctive, decreasing the risk of side effects, but they rapidly metabolize by proteases and allow short time activity in the body. Peptides' activity can be lengthened by incorporating modifications, such as non-natural and D-amino acids, cyclization and modifications at the N or C-terminus.
Therapeutic peptides also have some advantages compared to their protein counterparts. Biological therapeutics, which are generally proteins, have earned an increasing share of the pharmaceutical marketplace over the past few years. While biologics are often highly safe and effective, they must be produced in bioreactors, which use whole cells. Their purification and structural analysis is often complex and expensive. Further, biologics almost invariable must be injected. Peptides, on the other hand, can often be accessed chemically, and their purification and analysis is much simpler. There is also an increasing number of examples of orally active peptides, which make them more desirable for drug development.
Endogenous peptides have also been utilized for research and medical interventions. They can be monitored for diagnostic purposes, such as in the case of C-peptide, which is used to monitor insulin production and to help determine the cause of low blood sugar (hypoglycemia).
What are some examples of peptides?
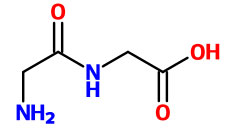
Synthetic peptides have been studied for over a century. The first synthetic peptide glycyl-glycine (see picture) was discovered by Emil Fischer in collaboration with Ernest Fourneau in 1901.
The first polypeptide (oxytocin - nine amino acid sequence) was synthesized by Vincent du Vigneaud in 1953.

The development of peptide therapeutics has made many advances over the years. Insulin was the first therapeutic protein to be introduced to treat insulin-dependent diabetes in the 1920s. It was initially isolated from bovine or porcine pancreases, but now human insulin is manufactured using genetically engineered E. coli. There are currently sixty FDA approved peptide drugs in the market, and pharmaceutical companies are increasingly interested in adding to that number. About 140 peptide drugs are in clinical trials and over 500 are in pre-clinical development.
How are peptides synthesized?
There are two major strategies for peptide synthesis: solution-phase peptide synthesis and solid-phase peptide synthesis (SPPS). SPPS is generally preferred because it does not require column purification after each coupling and deprotection step. Peptides are synthesized by coupling the carboxyl group or C-terminus of one amino acid to the amino group or N-terminus of another, lengthening the peptide from the C-terminus to the N-terminus. This process generally requires coupling agents and protecting groups. A variety of coupling agents and additives can be used during the coupling reaction, but the protecting groups for the terminal amino group is usually either an Fmoc (de-protected under basic conditions) or a Boc group (de-protected under acidic conditions).
With an understanding of peptides' functions and how they can by synthetically accessed, scientists can design, produce and utilize these powerful tools in research and clinical development.